|
7/31/2023 0 Comments Mass of hydrogen atom 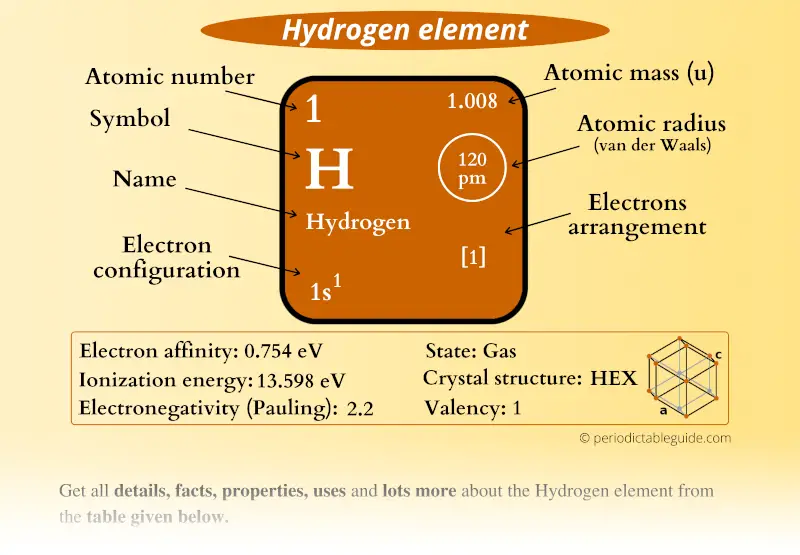 
because those electrons have similar average distances from the nucleus. All the wave functions that have the same value of n because those electrons have similar average distances from the nucleus. At room temperature and pressure, hydrogen exist as a diatomic gaseous molecule. The atomic weight of hydrogen is 1.00794 amu. All wave functions that have the same value of n are said to constitute a principal shell. Hydrogen is an s block element in the periodic table. This means that electrons with higher values of n are easier to remove from an atom. A negatively charged electron that is, on average, closer to the positively charged nucleus is attracted to the nucleus more strongly than an electron that is farther out in space. The relative atomic mass is worked out using the following formula, illustrated for two isotopes, where the abundances are given in percentage values.\]Īs\( \)n increases for a given atom, so does the average distance of an electron from the nucleus.  
In any sample of chlorine, 75 per cent of the atoms are 35 Cl and the remaining 25 per cent are 37 Cl. Therefore, Relative atomic mass average mass of an atom/mass of carbon-12 x (1/12) 1.0076632 u / 1 u 1. But the relative atomic mass of chlorine is not 36. Average mass of hydrogen (1.007825 u x 99.98) (2.014101 u x0.02 ) (1.007623 0.0000402) u 1.0076632 u The value of one twelfth of the mass of carbon-12 isotope is 1 u. For heavier hydrogen-like atoms it is closer to m. The relative atomic mass of an element is a weighted average of the masses of the atoms of the isotopes – because if there is much more of one isotope then that will influence the average mass much more than the less abundant isotope will.įor example, chlorine has two isotopes: 35 Cl and 37 Cl. In the hydrogen atom, in which the nucleus is just a proton, the ratio M / m is about 1836, so that 0.99946m. Calculating relative atomic mass from isotopic abundance The A r values also allow you to work out that three oxygen atoms have the same mass as two magnesium atoms.Ĭhlorine’s A r of 35.5 is an average of the masses of the different isotopes of chlorine. Bohr model of the Hydrogen Atom The atoms are generally made up of protons, neutrons and electrons. The first electron in helium has four quantum numbers of the first electron hydrogen. The molar mass of an element or molecule is the total mass in grams of all the atoms that comprise a mole of a certain. When the quantum number l0 there is only one value for m, which would be zero. In this scale, 1 atomic mass unit (amu) corresponds to 1.660539040 × 10 24 gram. It is expressed as a multiple of one-twelfth the mass of the carbon -12 atom, 1.992646547 × 10 23 gram, which is assigned an atomic mass of 12 units. They also tell you that hydrogen atoms have 12 times less mass than a carbon atom. The atomic molar mass of the hydrogen is 1.0078 u. atomic mass, the quantity of matter contained in an atom of an element. These values tell you that a magnesium atom has twice the mass of a carbon atom, and 24 times more mass than a hydrogen atom. Atoms with an A r that is more than this have a larger mass than a carbon atom.Atoms with an A r of less than this have a smaller mass than a carbon atom.Carbon is taken as the standard atom and has a relative atomic mass ( A r ) of 12. 1: The social security number subatomic-the proton. Lithium atoms have three protons, beryllium atoms have four, and so on. Next on the table is helium, whose atoms have two protons in the nucleus. Every hydrogen atom has one proton in its nucleus. Atoms have such a small mass it is more convenient to know their masses compared to each other. Hydrogen, at the upper left of the table, has an atomic number of 1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
